- Systemic dosing to deplete phagocytes of the RES, blood monocytes and other accessible phagocytes such as alveolar macrophages
- Local administration to deplete phagocytes in the area of interest
- Systemic and local dosing to maximize depletion and prevent repopulation in area of interest
| Contains: | mg/ml | mM | |
|---|---|---|---|
| Clodronate Disodium Salt | 5 | 17 | |
| L-α-Phosphatidylcholine | 18.8 | 24 | |
| Cholesterol | 4.2 | 11 | |
| Suspended in Phosphate Buffered Saline at pH 7.4 | |||
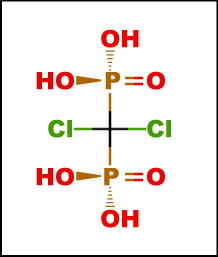
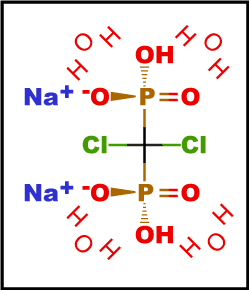
| Molecular Weight | 288.9 (Clodronate Disodium) 359.9 (Clodronate Disodium Tetrahydrate) |
| Formula | CH10Cl2Na2O10P2 (Tetrahydrate form) |
| Common names | Clodronate disodium; Dichloromethylene diphosphonate, disodium salt; DMDP; Cl2MDP |
| IUPAC | disodium tetrahydrate hydrogen [dichloro(hydrogen phosphonato)methyl]phosphonate |
| CAS Number | 22560-50-5 |
| Pubchem ID | 23724874 |
Other chemical properties and information on clodronate may be found at Pubchem, chemicalize.org and chemspider.com.
Clodronate belongs to a class of pharmaceutical compounds known as bisphosphonates which were originally developed for the treatment of bone malignancies. Their effect on bone is related to their ability to inhibit bone resorption by osteoclasts, thus bisphosphonates became frontline treatments for post-menopausal osteoporosis. Bisphosphonates have subequently been investigated for their potential efficacy in a wide variety of metastatic diseases and other conditions.
Clodronate is a non-nitrogen containing bisphosphonate ; the analogous nitrogen-containing subclass affect osteoclasts and other cells by a different biochemical mechanism (mevalonate pathway). The non-nitrogen-containing bisphosphonates, such as clodronate, are metabolized intracellularly to a non-hydrolyzable ATP analog which induces apoptosis by inibiting the ADP/ATP transport mechanism in mitochondria (see Mechanism of Depletion).
This bibliography references some of the many reviews written on bisphosphonates.
- A. Reszka, G. Rodan, Mechanism of action of bisphosphonates, Current Osteoporosis Reports, 1 (2003) 45–52.
- C.J. Rosen, C.R. Kessenich, Comparative Clinical Pharmacology and Therapeutic Use of Bisphosphonates in Metabolic Bone Diseases, Drugs, 51 (1996).
- D.L. Lourwood, The Pharmacology and Therapeutic Utility of Bisphosphonates, Pharmacotherapy: The Journal of Human Pharmacology and Drug Therapy, 18 (1998) 779–789.
- G.J. Yakatan, W.J. Poynor, R.L. Talbert, B.F. Floyd, C.L. Slough, R.S. Ampulski, J.J. Benedict, Clodronate kinetics and bioavailability, Clinical Pharmacology & Therapeutics, 31 (1982) 402–410.
- H. Ahn, J.M. Kim, K. Lee, H. Kim, D. Jeong, Extracellular acidosis accelerates bone resorption by enhancing osteoclast survival, adhesion, and migration, Biochemical and Biophysical Research Communications, 418 (2012) 144–148.
- F.H. Ebetino, A.-M.L. Hogan, S. Sun, M.K. Tsoumpra, X. Duan, J.T. Triffitt, A.A. Kwaasi, J.E. Dunford, B.L. Barnett, U. Oppermann, M.W. Lundy, A. Boyde, B.A. Kashemirov, C.E. McKenna, R.G.G. Russell, The relationship between the chemistry and biological activity of the bisphosphonates, Bone, 49 (2011) 20–33.
- R.G.G. Russell, Bisphosphonates: The first 40 years, Bone, 49 (2011) 2–19.
- Lipid composition is biosimilar egg-phosphatidylcholine and cholesterol in a 7:3 molar ratio
- Optimal size range (0.3-3 µm) for immediate phagocyte recognition and phagocytosis
- Extruded through 2 µm membranes to ensure that liposomes easily pass through lung capillaries
- Prepared and packaged under sterile conditions in stoppered glass vials with multidose adapters
- Batches prepared weekly to ensure maximal shelf-life after receipt by client
- Available as single product or in convenient Macrophage Depletion Kits
[custom_button text=”Order Now” title=”Order Now” url=”https://www.clodrosome.com/products/” size=”large” bg_color=”#002dcf” text_color=”#FFFFFF” align=”left” target=”_blank”][custom_button text=”Request Quotation” title=”Request Quotation” url=”https://www.clodrosome.com/order/request-quotation/” size=”large” bg_color=”#111111″ text_color=”#ffffff” align=”left” target=”_blank”][custom_button text=”Back to Products” title=”Back to Products” url=”https://www.clodrosome.com/products/” size=”medium” bg_color=”#333333″ text_color=”#ffffff” align=”right” target=”_blank”][custom_button text=”Back to Standard Clodrosome Kits” title=”Back to Standard Clodrosome Kits” url=”https://www.clodrosome.com/products/standard-clodrosome-reagents/standard-clodrosome-kits/” size=”medium” bg_color=”#333333″ text_color=”#ffffff” align=”right” target=”_blank”]